|
In addition, we monitored the change in ER ATP upon Ca 2+ release from the ER, and further evaluated the ER ATP status in response to varying cytosolic Ca 2+ concentrations. Specifically, we monitored real-time changes in ATP levels inside the ER lumen in response to well-characterized OxPhos and/or glycolysis inhibitors in living Chinese hamster ovary ( CHO), rat insulinoma INS1 and human Hela cells, at the single cell level using an ERAT-based FRET assay. Here, we studied ATP dynamics within the ER organelle in intact cells. A recent study revealed that the regulation of mitochondrial matrix ATP is highly dynamic and complex ( Depaoli et al., 2018). More recently, organelle specific ATP status determination was made possible with the genetically encoded FRET-based ATP reporter proteins targeted to select intracellular organelles, namely the mitochondrial localized AT1.03 and the ER localized ERAT4 probes ( Imamura et al., 2009 Vishnu et al., 2014). Although the level of ER ATP is suggested to impact protein secretion, this has not been demonstrated, nor have the factors that regulate ATP levels in the ER been clearly elucidated, although an association with ER Ca 2+ pool was suspected ( Vishnu et al., 2014 Klein et al., 2018). In fact, the level of ER ATP determines which proteins are able to transit to the cell surface ( Dorner et al., 1990 Dorner and Kaufman, 1994). The mammalian ER ATP transporter remained elusive until a recent publication identified SLC35B1/AXER as the putative mammalian ER ATP transporter ( Klein et al., 2018).ĮR ATP is essential to support protein chaperone functions for protein folding, such as BiP/GRP78, and trafficking ( Dorner et al., 1990 Braakman et al., 1992 Dorner and Kaufman, 1994 Wei et al., 1995 Rosser et al., 2004). For example, only one report described the genetic identification of an ATP transporter ER-ANT1 in Arabidopsis which is restricted to plants and its deletion caused a disastrous plant phenotype, characterized by drastic growth retardation and impaired root and seed development ( Leroch et al., 2008). Other than that, little is known about how ATP gets into the ER in a living cell or whether/how ATP consumption is regulated in the ER lumen. A membrane protein, ‘ ER-ANT’, is involved in the ATP translocation across the ER membrane, of which the biochemical properties are analogous to the mitochondrial Adenosine Nucleotide Transporter ( ANT) ( Clairmont et al., 1992 Shin et al., 2000). Despite its heavy demand for ATP to facilitate protein folding and trafficking, the ER is not known to possess an independent ATP regeneration machinery. ATP regeneration from ADP takes place in mitochondria mainly through OxPhos, and in the cytosol through glycolysis. Universally, ATP is the single most important energy-supplying molecule in every form of life. an ‘ anti-Warburg’ effect.Įnergy supply is a fundamental requirement for all cells to perform their biochemical functions. These findings suggest that ATP usage in the ER may increase mitochondrial OxPhos while decreasing glycolysis, i.e. Furthermore, the ATP level in the ER is readily depleted by oxidative phosphorylation ( OxPhos) inhibitors and that ER protein misfolding increases ATP uptake from mitochondria into the ER. 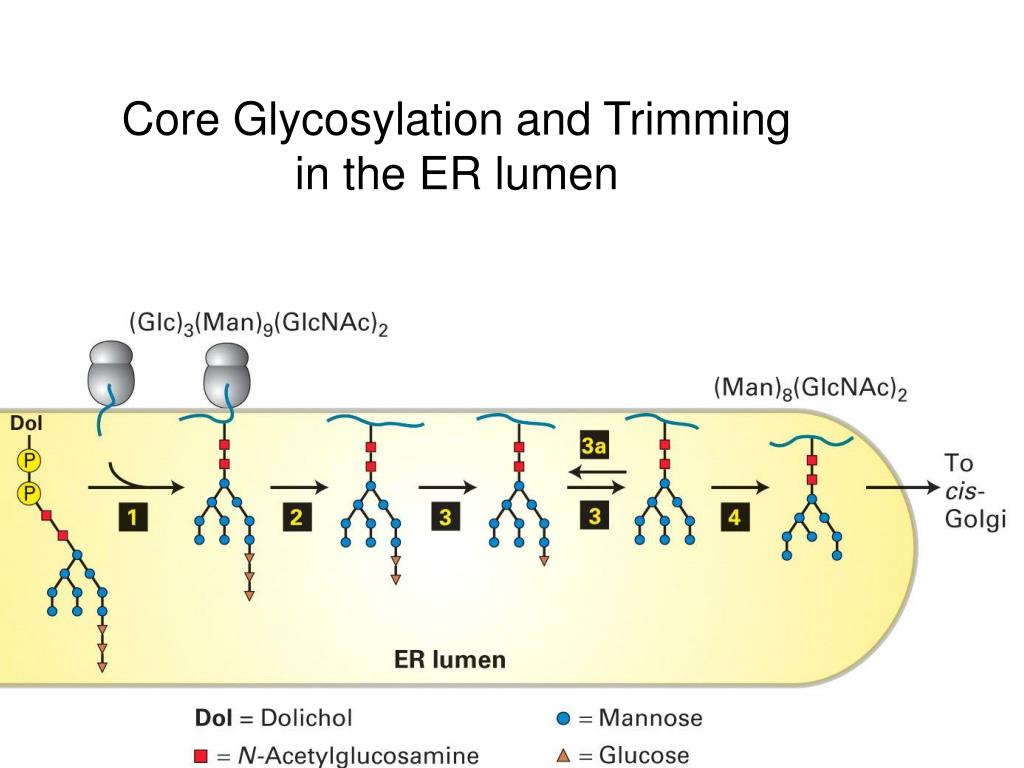 
We propose that under physiological conditions, increases in cytosolic Ca 2+ inhibit ATP import into the ER lumen to limit ER ATP consumption. Significantly, we show that mitochondria supply ATP to the ER and a SERCA-dependent Ca 2+ gradient across the ER membrane is necessary for ATP transport into the ER, through SLC35B1/AXER. Here, using three commonly used cell lines (CHO, INS1 and HeLa), we report that ATP enters the ER lumen through a cytosolic Ca 2+-antagonized mechanism, or CaATiER ( Ca 2+- Antagonized Transport into ER). However, little is known about how this vital ATP transport occurs across the ER membrane. The endoplasmic reticulum ( ER) imports ATP and uses energy from ATP hydrolysis for protein folding and trafficking.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
